Chemistry
Chemical Automation
 Chemical Automation is the main component of Water Treatment. Chemical Automation refers to the automatic monitoring and control of the sanitizer and pH levels in water to maintain clean and safe water in order to prevent corrosion, scaling, growth of germs and bacteria (such as E. Coli and Pseudomonas) and ….. closure by the Health Department !
Chemical Automation is the main component of Water Treatment. Chemical Automation refers to the automatic monitoring and control of the sanitizer and pH levels in water to maintain clean and safe water in order to prevent corrosion, scaling, growth of germs and bacteria (such as E. Coli and Pseudomonas) and ….. closure by the Health Department !
By constantly monitoring the pH of the water and the activity of the sanitizer, Chemical Automation makes it possible to maintain clean and safe water at all times with a significant reduction in the cost of chemicals, maintenance and repairs.
It is now recognized as a must for all commercial and public pools and spas, as well as for quality residential installations.
Sanitizer Control
The activity of the sanitizer is measured with a special electrode called an ORP or Redox electrode. Both of these terms stand for Oxidation-Reduction Potential. ORP does not measure directly the amount of sanitizer in the water but rather its activity, which makes it more valuable to proper water sanitation.
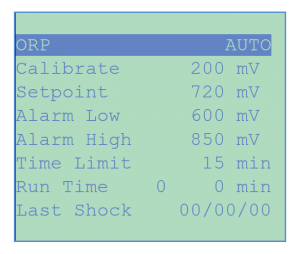 The ORP menu readings are used to monitor the activity of the sanitizer: chlorine, bromine and or ozone. ORP is measured in millivolts (mV) and provide a superior index of bacteriological water quality because it monitors the activity of the sanitizer rather than just its quantity as with conventional PPM (parts per million) test kits.
The ORP menu readings are used to monitor the activity of the sanitizer: chlorine, bromine and or ozone. ORP is measured in millivolts (mV) and provide a superior index of bacteriological water quality because it monitors the activity of the sanitizer rather than just its quantity as with conventional PPM (parts per million) test kits.
ORP MENU includes a selection of control modes (Off, Manual, Auto or Timer), sensor calibration (1, 2 or 3 points), set-point level, high and low alarms, shocking and chemical saver programs. It also displays the run time for the current feed event and cumulative run time. Shock treatment can be initiated manually or under programmable weekly or multi-weekly schedules.
…
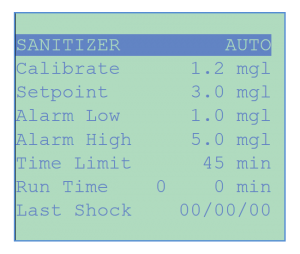
The SANITIZER menu is used to control the chlorine or bromine level in terms of sanitizer concentration (parts per million or milligrams/liter), obtained by conversion from ORP and pH values using a proprietary algorithm or from an amperometric sensor.
…
The sanitizer concentration readings can be calibrated to account for the effect of cyanuric acid stabilization. Super-chlorination can be initiated manually or under programmable weekly or multi-weekly schedules.
…
pH Control
 pH represents the acidity or basicity of the water on a logarithmic scale that represent the concentration of hydrogen ions H+ in the water. pH is very important to water chemistry because the hydrogen ions are small and very active and therefore affect most chemical reactions in water.
pH represents the acidity or basicity of the water on a logarithmic scale that represent the concentration of hydrogen ions H+ in the water. pH is very important to water chemistry because the hydrogen ions are small and very active and therefore affect most chemical reactions in water.
pH values range from 0 to 14, with 7 being neutral. For instance a neutral pH of 7 represents a H+ ion concentration of 10(-7) or 1/10,000,000, while a basic pH of 8 represents a concentration of only 10(-8) or 1/100,000,000.
The pH scale is logarithmic. When the pH value increases by one unit, i.e. from 7 to 8, the acidity of the water decreases tenfold. Small changes in pH therefore have large effects and this is why close pH control is critical to water maintenance.
The ideal pH range for water treatment is 7.4 to 7.6. Below 7.4, the water is too acidic. This causes eye irritation and corrosion of the equipment (pump, filter, plaster, etc). Above 7.6, the water is too alkaline. This causes eye irritation plus cloudiness, scaling and stains.
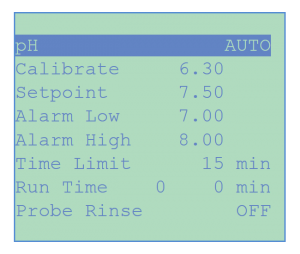 The pH menu is used in the same way to select the pH control parameters using either acid or base feed or both. It also includes an Acid Wash program for periodic injection of an acid rinse solution to clean the heads of the sensors.
The pH menu is used in the same way to select the pH control parameters using either acid or base feed or both. It also includes an Acid Wash program for periodic injection of an acid rinse solution to clean the heads of the sensors.
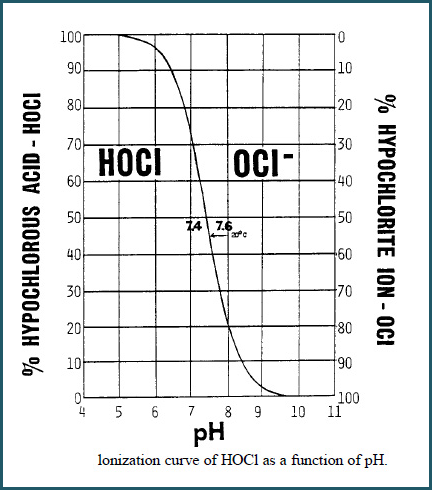
Most important, pH affects the dissociation, and therefore the effectiveness, of Hypochlorous Acid HOCl, the most active sanitizer form of Free Chlorine, as shown in the curve below.
At a pH of 7.5, Free Chlorine is about 50% HOCl . At higher pH values, HOCl dissociates into the ionic form OCL- which is a less active sanitizer.
The pH of the water can increase due to the addition of alkaline (basic) chemicals such as liquid chlorine (Sodium Hypochlorite NaOCl), dry chlorine (Calcium Hypochlorite Ca[OCl]2) or make-up water. It can decrease with the addition of Chlorine Gas (Cl2), body perspiration, acid rain or make-up water.
Acids are used to decrease the pH of the water, usually liquid muriatic acid (Hypochloric Acid HCl), dry acid (Sodium Bisulfate NaHSO4) or Carbon Dioxide CO2 gas. Bases like caustic soda (Sodium Hydroxide NaOH) or dry soda are used to raise the pH.
The addition of pH correction chemicals used to be done manually or with a chemical feeder. It is now done on demand with automatic controllers.
A chemical controller monitors the pH of the water with an electronic sensor consisting of a glass pH electrode.
The pH sensor readings vary from 0 mV for a neutral pH of 7 to about -60 mV for a pH of 8. For the recommended pH of 7.4, the reading is about -25 mV. It should be recognized however that the pH readings vary slightly from electrode to electrode. This is why pH testers and controllers always include a pH calibration adjustment. Calibration can be made with a Phenol Red test kit.
Principles of Water Treatment
Water treatment for swimming pools, spas and industrial applications requires the combination of a number of physical and chemical processes, including pumping, filtration, chemical treatment, heating and water replacement.
The primary objectives of water treatment are:
- Maintaining clean and safe water that meets the bacteriological and physiological requirements of state and local Health Departments,
- Protecting the equipment from corrosion or scaling caused by the aggressiveness of water and its constituents.
To meet these objectives, the CHEMTROL® Programmable Controllers incorporates all the monitoring, control and communication functions into a single integrated command center designed around a powerful microprocessor. The integrated Main Screen display enables the operator to supervise all the process functions at a glance and to quickly respond to any changing condition.

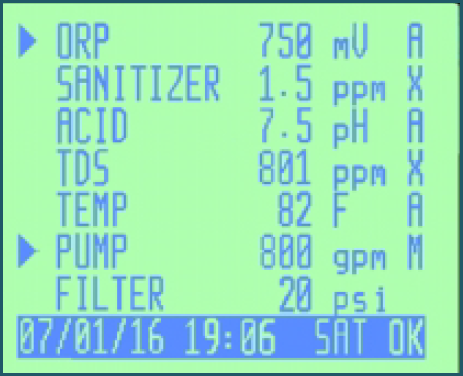
The CHEMTROL® Programmable controllers operate like a computer with full- screen displays and direct access to all menus and submenus. The front panel keypad is used to move up and down each menu and enter or exit the submenus. All operating functions can be easily modified from the keypad, including sensor calibration, control set-points, alarm levels and the programs for super-chlorination, sequential backwashing, chemical saving and energy saving.
Implementation
Due to the processing equipment is made by different manufacturers and controlled individually, installation and operation of any automated system might be complicated and inefficient. 
Nowadays, most countries require an automatic process for sanitizer and pH control in all commercial, semi-public and public aquatic facilities. As a minimum, this refers to an automatic chemical feeder such as a pump or an erosion-feeder or some other kind of fixed rate dispenser with or without a timing device.
CHEMTROL® controllers have kept millions of bathers and the general public safe, ensuring consistent disinfection under varying conditions and regulations. This is especially critical when mandatory public health obligations must be met. And with all control, monitoring and communication functions accessible from a single integrated command center, you have complete peace of mind of your water treatment control and chemical automation needs.
